Students can Download Chapter 10 The s Block Elements Questions and Answers, Plus One Chemistry Chapter Wise Questions and Answers helps you to revise the complete Kerala State Syllabus and score more marks in your examinations.
Kerala Plus One Chemistry Chapter Wise Questions and Answers Chapter 10 The s Block Elements
Plus One Chemistry The s Block Elements One Mark Questions and Answers
Question 1.
The element placed at the bottom of the alkali metal family is expected to
a) Have maximum ionisation enthalpy
b) Be the least reducing agent
c) Be the least electropositive element
d) Be the most easily ionisable
Answer:
d) Be the most easily ionisable
Question 2.
Which of the following have lowest thermal stability?
a) Li2CO3
b) Na2CO3
c) K2O3
d) Rb2CO3
Answer:
a) Li2CO3
![]()
Question 3.
The chemical formula of Plaster of Paris is
a) Ca2SiO4.½H2O
b) Ca2SiO4.2H2O
c) CaSO4.½H2O
d) CaSO4.2H2O
Answer:
c) CaSO4.½H2O
Question 4.
A mixture of NaOH and CaO is known as _________ .
Answer:
Soda lime
Question 5.
Which of the following is least soluble in water?
a) BeSO4
b) BaSO4
c) CaSO4
d) SrSO4
Answer:
BaSO4
Question 6.
Pick out the odd one and write reason for it.
Ca(OH)2, Mg(OH)2, Ba(OH)2, Be(OH)2 Sp
Answer:
Be(OH)2. The others hydroxides are basic but Be(OH)2 is amphoteric.
Question 7.
The stability of alkaline earth metal carbonate
Answer:
Increases from Be to Ba
Question 8.
Mg2C3 on hydrolysis gives _________
Answer:
Propyne
![]()
Question 9.
The raw materials required for the manufacture of cement clinker are
Answer:
limestone & clay
Question 10.
A sodium potassium alloy is used as a _________ .
Answer:
coolant in nuclear reactor.
Question 11.
Li2CO3 decomposes at a lower temperature whereas Na2CO3 at highertemperature _________ .
Answer:
Li+ is very small
Plus One Chemistry The s Block Elements Two Mark Questions and Answers
Question 1.
Choose the false statements and rewrite them.
a) Alkali metals possess both +1 & +2 oxidation states.
b) Lithium is found to be the strongest reducing agents among the alkali metals.
c) Manufacturing of rayon is known as viscose process.
d) Washing soda is used to remove temporary hardness of water.
Answer:
a) False. Alkali metals possess only +1 oxidation state.
b) True
c) True
d) False. Washing soda is used to remove the permanent hardness.
Question 2.
a) Write the scientific name of slaked lime.
b) What is its role in white wash?
c) What do you mean by slaking of lime?
d) What happens when slaked lime is treated with dry chlorine?
Answer:
a) Calcium hydroxide
b) It is disinfectant hence it is used in white wash.
c) Calcium hydroxide is obtained by adding water to quick lime. This process is called slaking of lime.
d) Calcium hydroxide reacts with dry chlorine to form calcium hypochlorite, a constituent of bleaching powder.
2Ca(OH)2 + 2Cl2 → CaCl2 + CaOCl2 + 2H2O
Question 3.
1. Which metal is present in chlorophyll?
2. What is the role of calcium in our body?
Answer:
1. Mg
2. About 90% of body calcium is present in bones and teeth. It also plays important roles in neuromuscular function, intemeuronal transmission, cell membrane integrity and blood coagulation.
![]()
Question 4.
A compound is used as drying agent as such or as soda lime with NaOH and it is used on a very large scale as a building material.
a) Which is this compound?
b) How can we prepare this compound?
c) What are the properties of this compound?
Answer:
a) Calcium oxide
b) It can be prepared by heating lime stone in a rotary kiln at 1070-1270 K.
c) Pure calcium oxide is an amorphous white solid of very high melting point. It is sparingly soluble in water. Calcium oxide readily absorbs moisture and carbon dioxide.
Question 5.
Listen the chemical reaction,
![]()
- Write the common name of the reactant.
- What is the temperature corresponding to A?
- What is B (product)? Write its chemical formula.
Answer:
- Gypsum
- 393 K
- Plaster of Paris (CaSO4. ½H2O) or (CaSO4)2.H2O
Question 6.
Explain with the help of chemical equations what happens when
- lime stone is heated?
- water is dropped on quick lime?
- gypsum is heated to 393 K?
Answer:
1. When lime stone is heated, it is converted into CaO and CO2.
![]()
2. When water is dropped on quick lime calcium hydroxide is formed.
CaO + H2O → Ca(OH)2
3. When gypsum is heated to 393 K, Plaster of Paris is obtained.
![]()
Question 7.
Alkali metal halides are all high melting, colourless crystalline solids.
- Write any other physical property of alkalimetal halides.
- How alkali metal halides are prepared?
Answer:
1. All the alkali metal halides are soluble in water except LiF. They have high negative enthalpies of formation. The melting and boiling points always follow the trend: fluoride > chloride > bromide > iodide.
2. Alkali metal halides are prepared by the reaction of the appropriate oxide, hydroxide or carbonate with aqueous hydrohalic acid.
![]()
Question 8.
What is Plaster of Paris? How is it prepared? What is its use?
Answer:
The chemical formula of plaster of Paris is (CaSO4)2.H2O.
It is prepared by heating gypsum at 393 K.
![]()
It is a hemihydrate of calcium sulphate.
It is a white powder. On mixing with an adequate quantity of water it forms a plastic mass that gets into a hard solid in 5 to 10 minutes.
Plaster of Paris is used
- for making moulds for pottery, ceramics, etc.
- for making models, statues and decorative materials.
- for immobilising the affected part of organ where there is a bone fracture or sprain.
Question 9.
Give the biological importance of Na and K.
Answer:
Biological importance of sodium: The Na+ ions participate in the transmission of nerve signals, in regulating the flow of water across cell membranes and in the transport of sugars and amino acids into cells. Biological importance of potassium: The K+ ions activate many enzymes, participate in the oxidation of glucose to produce ATP and, with sodium, are responsible for the transmission of nerve signals.
Question 10.
1. Name the element showing anomalous behaviour in group 2.
2. Give reason forthis anomalous behaviour,
3. List any two similarities between Be and Al.
Answer:
1. Be
2. Be has exceptionally small atomic and ionic size, it has high ionisation enthalpy, it cannot exhibit coordination number more than four as in its valence shell there are only four orbitals. There are no d-orbitals.
2. i) Both Be and Al are not readily attacked by acids because of the presence of an oxide film on the surface of the metal.
ii) The chlorides of both Be and Al have Cl– bridged chloride structure in vapour phase.
Plus One Chemistry The s Block Elements Three Mark Questions and Answers
Question 1.
A compound of calcium is used for immobilising the fractured bones of body.
- Write down the common name and molecular formula of the compound.
- Which property of the compound helps to make plaster?
- What do you mean by dead burnt plaster? How does it form?
Answer:
1. Plaster of Paris (CaSO4. ½H2O) or (CaSO4)2.H2O
2. When we add water to Plaster of Paris, there is slight increase in volume and this helps Plaster of Paris to take the shape of any mould in which it is present.
3. When Plaster of Paris is heated above 393 K, Plaster of Paris loses water of crystallization to form anhydrous calcium sulphate, CaSO4. It is known as dead burnt plaster which does not set in presence of water.
Question 2.
Lithium is group 1 element. It shows some similarities with group 2 element magnesium.
1. Write the name of the relationship.
2. Explain this relationship.
3. What is the reason for this relationship?
4. Give other example for this relationship.
Answer:
1. Diagonal relationship.
2. The first element of alkali group shows some similarities with the second element of the second group, which is diagonally present. This relation is called diagonal relationship.
3. Diagonal relationship is due to the following reasons:
- Similarity in their polarising powers due to similar charge/radius ratio.
- Almost similar electronegativity of the 2 elements.
4.
- Berylium and Aluminium
- Boron and Silicon
![]()
Question 3.
Mentionafewimportarrtusesofthefollowing compounds.
- Epsom salt
- Marbl
- Sodium hydroxide
Answer:
1. Epsom salt:
It is used as a mordant in dyeing. Used in medicine in purgative. Used in tanning industry.
2. Marble:
It is used to make quick lime, cement etc. Used as a raw material in the Solvay process. Used as a building material.
3. Sodium hydroxide:
It is used as a laboratory reagent. Used in petroleum refining. Used in viscose process.
Question 4.
Cement is a complex mixture of silicates and aluminates.
1. What is the function of gypsum in cement?
2. Explain setting of cement.
Answer:
1. The purpose of adding gypsum is only to slow down the process of setting of the cement so that it gets sufficiently hardened.
2. When the cement is mixed with water, it forms a gelatinous mass which sets slowly to a hard mass having 3 dimensional network structure with -Si- O-Si and -Si-O-AI- chains. This is an exothermic process and is called setting of cement.
Question 5.
The alkali metals and their salts impart characteristic colour to an oxidising flame.
1. What is the reason for this?
2. Give the flame colour of Na and K.
3. Name the alkali metal which imparts crimson red colour to the oxidizing flame.
Answer:
1. When the heat energy is supplied to alkali metal or its salt, the electrons are excited to higher energy levels. When these excited electrons come back to their original energy levels, they emit radiations which fall in the visible region of the electromagnetic spectrum and they appear coloured.
2. Na-Yellow, K-Violet
3. Li
Question 6.
a) How will you prepare Ca(OH)2 and CaCO3 from quicklime.
b) Give any two uses of quick lime.
Answer:
a) By adding water to quick lime we can prepare Ca(OH)2. This process is called slaking.
CaO + H2O → Ca(OH)2
When CO2 is passed thruogh lime water we can prepare CaCO3.
Ca(OH)2 + CO2 → CaCO3 + H2O
b) 1. As an important primary material for manufacturing cement.
2. In the manufacture of Na2CO3 from NaOH.
Question 7.
Lithium shows similarities in properties with Magnesium.
a) Namethe above phenomenon.
b) Give any two similarities of Lithium with Magnesium.
c) How is bleaching powder prepared?
Answer:
a) Diagonal relationship
b) 1) Both Lithium and Magnesium are harder and lighter than other elements in the respective groups.
2) The oxides of both (Li2O and MgO) do not combine with excess O2 to give any superoxide.
c) It is prepared by passing Cl2 gas through dry slaked lime.
2Ca(OH)2 + 2Cl2 → CaCl2 + CaOCl2 + 2H2O
Plus One Chemistry The s Block Elements Four Mark Questions and Answers
Question 1.
1. What is the name of the method that is used in manufacturing of sodium hydroxide?
2. Explain the method.
3. Write the equations of the reactions involved in this process.
Answer:
1. Castner-Kellner method
2. The Castner-Kellner cell consists of a large tank. The bottom of the tank is filled with mercury. The tank is made into 3 compartments by 2 partitions. The outer compartments are filled with NaCl solution and the middle compartment is filled with a very dilute solution of NaOH. The mercury layer of the bottom of cell in the outer compartments acts as cathode and the mercury in the middle compartments acts as anode. As a result of electrolysis Na and Cl2 are produced in the outer compartments. Na gets coated with mercury, and sodium amalgam is produced in the outer compartments. Due to the action of eccentric wheels at the bottom of the tank, the tank can be tilted. As a result of this Na is carried into the middle compartment and is then allowed to react with water in the middle compartment. As a result of this reaction NaOH is produced. When the concentration of NaOH in the middle compartment is raised to 40%, it is taken out from the tank. On cooling, crystals of NaOH separate out.
3. NaCl → Na+ + Cl–
At cathode: Na+ + e– → Na
Na + Hg → Na/Hg (Sodium amalgam)
At anode: Cl– → Cl + e–
Cl + Cl → Cl2 (g)
Question 2.
Match the following:

Answer:
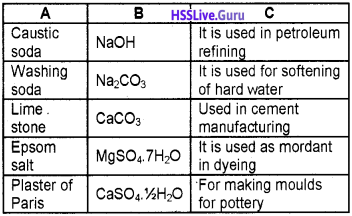
Question 3.
A piece of metallic sodium is added to liquid ammonia.
- What is the observation?
- What is the reason for this?
- What happens when the solution is kept for some time?
- What happens if the solution is concentrated?
Answer:
- The solution turns into blue.
- The blue colour of the solution is due to the presence of solvated electrons.
- On standing, the solution slowly liberates hydrogen resulting in the formation of amide.
- When the concentration of the solution increases the colour turns bronze and the solution becomes diamagnetic.
Question 4.
1. Some compounds of sodium and calcium are given below. Match them.

2. On passing CO2 through lime water, milkiness appears. On further passing CO2 milkiness disappears. What is the Chemistry behind it?
Answer:
1.

2. When CO2 is passed through lime water it turns milky due to the formation of insoluble calcium carbonate.
Ca(OH)2 + CO2 → CaCO3 +H2O
When CO2 is passed through lime water for a long time, it becomes colourless due to the formation of water soluble calcium bicarbonate.
CaCO3 + H2O + CO2 → CaOHCO3)2
Question 5.
Cement is an important building material employed in different kinds of construction works.
- What are the major raw materials for making cement?
- How is cement prepared?
- What are the important ingredients of Portland cement?
- Explain the Chemistry involved in the setting of cement.
Answer:
1. Limestone, Clay
2. When clay and lime are strongly heated together they fuse and react to form cement clinker. This clinker is mixed with 2-3% by weight of gypsum to form cement.
3. Dicalcium silicate (Ca2SiO4) – 26%, tricalcium silicate (Ca3SiO5) – 51% and tricalcium aluminate (Ca3Al2O6) – 11%
4. When mixed with water, the setting of cement takes place to give a hard mass. This is due to the hydration of the molecules of the constituents and their rearrangement.
Question 6.
1. Match the following :

2. Study the list of elements given below:
Na, Mg, Li, B, C
Select the pairs showing diagonal relationship.
3. Write the chemical equation showing the reaction of sodium metal with water.
Answer:
1.
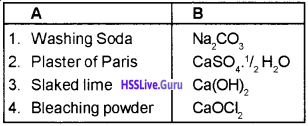
2. Li, Mg
3. 2Na + 2H2O → 2NaOH + H2
Question 7.
a) Arrange the following compounds in the increasing order of solubility in water:
i) Mg(OH)2, Be(OH)2, Ba(OH)2, Ca(OH)2, Sr(OH)2
ii) SrSO4, MgSO4, BeSO4, BaSO4, CaSO4,
b) i) Write the difference between lime water and milk of lime.
ii) Complete the following reaction.
![]()
Answer:
a) i) Be(OH)2 < Mg(OH)2< Ca(OH)2< Sr(OH)2< Ba(OH)2
ii) BaSO4 < SrSO4< CaSO4< MgSO4< BeSO4
b) i) A clear solution of calcium hydroxide in water is called lime water. A suspension of calcium hydroxide in water is called milk of lime,
![]()
Plus One Chemistry The s Block Elements NCERT Questions and Answers
Question 1.
Explain why alkali and alkaline earth metals cannot be obtained by chemical reduction methods? (2)
Answer:
Alkali and alkaline earth metals are themselves very strong reducing agents and reducing agents stronger than them are not easily available. Therefore, these metals cannot be obtained by chemical reduction methods.
Question 2.
Why are potassium and caesium, rather than lithium used in photoelectric cells? (2)
Answer:
Potassium and caesium have much lower ionisation enthalpy than that of lithium. As a result, these metals on exposure to light, lose electrons much more easily than lithium. Hence, potassium and caesium rather than lithium are used in photoelectric cells.
![]()
Question 3.
When an alkali metal dissolves in liquid ammonia the solution can acquire different colours. Explain the reasons for this type of colour change. (2) Answer:
When an alkali metal is dissolved in liqiud ammonia it produces a blue coloured conducting solution due to formation of ammoniated cation and ammoniated electron as given below:
![]()
When the concentration is above 3 M, the colour of solution is copper-bronze. This colour change is because the concentrated solution contains clusters of metal ions and hence possess metallic lustre.
Question 4.
Beryllium and magnesium do not give colour to flame whereas other alkaline earth metals do so, why? (2)
Answer:
Due to the small size, the ionisation enthalpies of Be and Mg are much higher than those of other alkaline earth metals. This means that the valence electrons in beryllium and magnesium are more tightly held by the nucleus. Therefore, they need large amount of energy for excitation of electrons to higher energy levels. Since such a large amount of energy is not available in bunsen flame, therefore, these metals do not impart any colour to the flame.
Question 5.
Potassium carbonate cannot be prepared by Solvay process. Why? (2)
Answer:
Potassium carbonate cannot be prepared by Solvay process because potassium biocarbonate being highly soluble in water does not get precipitated in carbonation tower when CO2 is passed through a concentrated solution of KCI saturated with ammonia.
![]()
Question 6.
Why Li2CO3 decomposes at lower temperature whereas Na2CO3 at higher temperature? (2)
Answer:
Li+ ion is smaller in size. It forms more stable lattice with smaller anion oxide, O2- than with CO32- ion. Therefore, Li2CO3decomposes into Li2O at lower temperature on the other hand, Na+ ion being larger in size forms more stable lattice with larger anion CO32- than with O2- ion. Therefore, Na2CO3 is quite stable and decomposes into Na20 at very high temperature.
Question 7.
How would you explain? (3)
1. BeO is insoluble but BeSO4 is soluble in water.
2. BaO is soluble but BaSO4 is insoluble in water.
3. Lil is more soluble than Kl in ethanol.
Answer:
1. BeO has higher lattice enthalpy than hydration enthalpy and hence is insoluble in water. BeSO4 on the contrary, is soluble because the lattice enthalpy is less due to bigger sulphate ion.
2. In barium oxide, BaO, due to larger size of barium ion the lattice enthalpy is less than hydration enthalpy and hence it is soluble in water. On the other hand, in BaSO4 dueto largersize of barium as well as sulphate ion, the magnitude of hydration enthalpy is much smallerthan the lattice enthalpy and hence it is insoluble in water.
3. In Kl the chemical bond is ionic in character. On the other hand, due to small size of lithium ion and its high polarising power, the bond in Li is predominantly covalent in character. Hence, Li is more soluble than Kl in ethanol.
Question 8.
Which of the alkali metals is having least melting point? Justify. (2)
a) Na
b) K
c) Rb
d) Cs
Answer:
d) Cs.
As the atomic size of the metal increases, the strength of metallic bonding decreases and hence its melting point decreases. Since the size of Cs is the largest, therefore its melting point is the lowest.